What To Know
- Osmosis is the movement of water molecules across a semipermeable membrane from a region of high water concentration (or low solute concentration) to a region of low water concentration (or high solute concentration).
- Both diffusion and osmosis describe how molecules move from an area of high concentration to an area of low concentration, but they differ in the substances involved.
Osmosis Examples: Meaning, Mechanism, and Real-Life Applications
Understanding osmosis examples is essential for anyone studying biology, physiology, or chemistry. Osmosis plays a critical role in the survival of plants and animals, influencing how water molecules move across biological systems. It explains how cells maintain balance, how plants absorb water from the soil, and how medical treatments regulate fluids in the body.
This article explores the osmosis definition, explains the process of osmosis, highlights examples of osmosis, and shows the significance of osmosis in biological systems.
Osmosis Definition: Understanding the Movement of Water Across a Membrane
Osmosis is the movement of water molecules across a semipermeable membrane from a region of high water concentration (or low solute concentration) to a region of low water concentration (or high solute concentration).
In simple terms, the passage of water occurs across a semi-permeable membrane until equilibrium is reached.
Key components of the osmosis process include:
- Solvent: Usually water
- Solute: A substance that can dissolve in water
- Membrane: A permeable barrier that allows water but not solute particles to pass
In biological systems, the cell membrane acts as the semipermeable membrane through which water across the membrane moves.
During this process:
- Water molecules move toward the side of the membrane with higher solute concentration.
- The net movement of water continues until equilibrium occurs.
- This net flow of water balances the solute concentration on both sides.
Thus, osmosis occurs whenever water moves across a semipermeable membrane due to differences in concentration of solute.
Diffusion and Osmosis: Understanding Their Relationship
Both diffusion and osmosis describe how molecules move from an area of high concentration to an area of low concentration, but they differ in the substances involved.
Diffusion
Diffusion refers to the movement of any substance (gas, liquid, or dissolved particles) from high concentration to lower concentration.
Examples include:
- Oxygen diffuse into cells
- Carbon dioxide diffusing out of cells
Osmosis
Osmosis, on the other hand, is a specific type of diffusion involving only water molecules.
The movement of water across a semipermeable membrane is driven by differences in water concentration or solute concentration.
Thus:
- Diffusion and osmosis both involve net movement.
- Osmosis and diffusion both occur until equilibrium is reached.
- However, osmosis is essential for maintaining cellular balance.
Osmosis Diagram: Visualizing the Osmosis Process
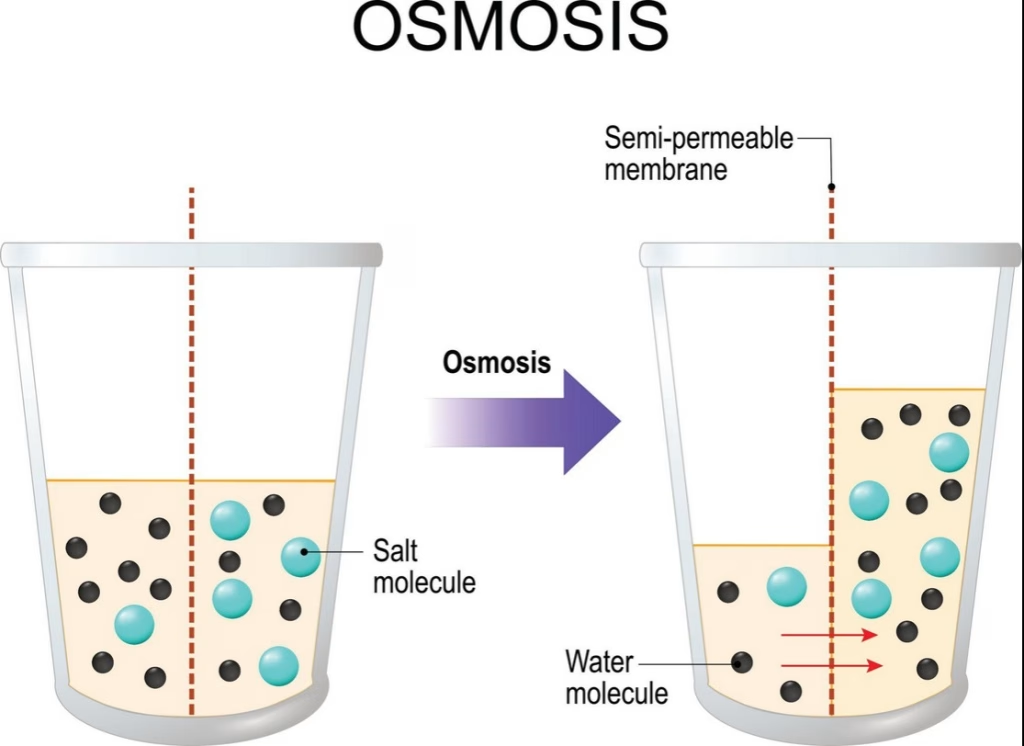
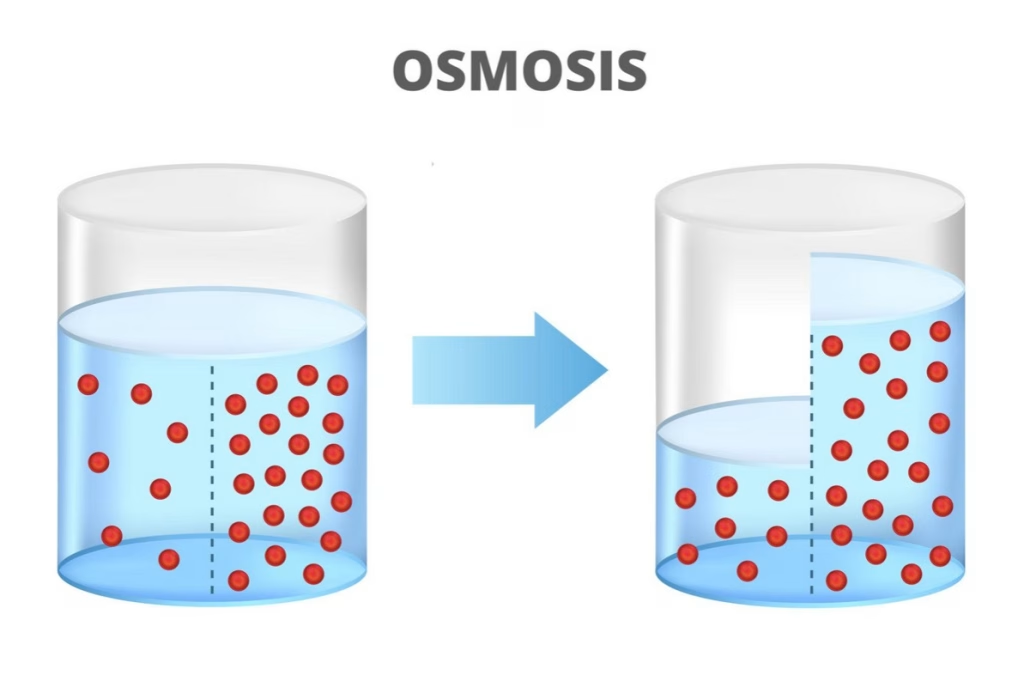
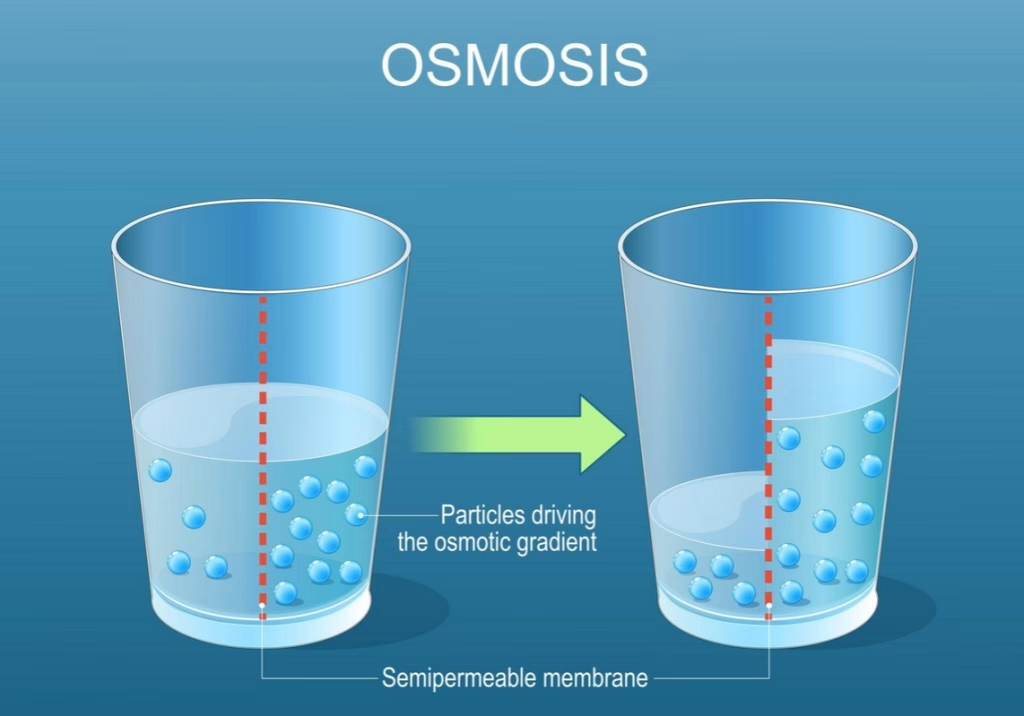
A typical osmosis diagram illustrates:
- A semi-permeable membrane separating two solutions
- A region of lower solute concentration and high water concentration
- A region of higher solute concentration and low water concentration
During the osmosis process:
- Water molecules move from the region of high water to the region of low water.
- The movement of water across the membrane creates osmotic pressure.
- Eventually, the net movement of water slows as the system approaches equilibrium.
These diagrams help students visualize how water flows across a membrane in biological systems.
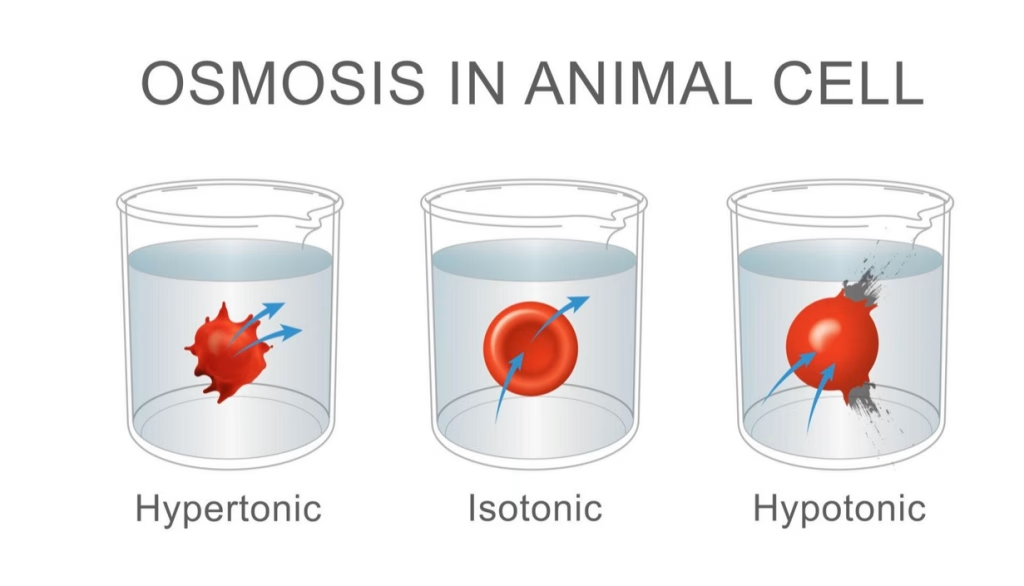
Types of Solutions in Osmosis: Hypertonic, Hypotonic, and Isotonic
The direction of water moves depends on the types of solutions surrounding the cell.
1. Hypotonic Solution
A hypotonic solution has low solute concentration and high water concentration.
Effects on cells:
- Water moves into the cell
- The animal cell may swell
- The flow of water increases internal pressure
For example, when a red blood cell is placed in a hypotonic solution, water flows into the cell.
2. Hypertonic Solution
A hypertonic solution has high solute concentration and low water concentration.
Effects on cells:
- Water moves out of the cell
- The cell may shrivel
For instance:
- A red blood cell placed in a hypertonic solution loses water
- The red blood cells shrink due to net flow of water out of the cell
3. Isotonic Solution
An isotonic solution has equal solute concentration inside and outside the cell.
In this case:
- The net movement of water is balanced
- Water molecules move in both directions
- The net flow of water equals zero
Thus, the cell remains stable because inside and outside concentrations are the same.
Need Help Writing a Biology Paper on Osmosis?
Understanding complex topics like osmosis, diffusion, cell membranes, and solute concentration can be challenging, especially when writing research papers, lab reports, or assignments. If you need expert assistance, IvyResearchWriters.com is here to help.
Examples of Osmosis in Everyday Life and Biology
Understanding osmosis examples helps explain many natural and biological processes.
1. Plant Roots Absorbing Water
Plants absorb water from the soil through root hairs.
Here:
- Soil water has high water concentration
- Root cells have higher solute concentration
Therefore, water moves into the cell through the cell membrane via the process of osmosis.
2. Osmosis in Red Blood Cells
The behavior of red blood cell membranes demonstrates clear osmosis examples.
- In a hypotonic solution, water molecules move into the cell.
- In a hypertonic solution, water leaves and the cell shrivel.
- In an isotonic solution, the net movement of water is balanced.
These changes illustrate how osmosis occurs across the cell membrane.
3. Food Preservation Using Salt
Salt creates a high solute concentration environment.
When microorganisms encounter salt:
- Water moves out of the cell
- The cell dehydrates
- Growth stops
This osmosis example explains why salt preserves food.
4. Kidney Function in Humans
The kidneys regulate body fluids through osmosis and diffusion.
During filtration:
- Water across kidney membranes moves according to solute concentration.
- Osmotic pressure helps maintain fluid balance in the body.
This mechanism is essential in physiology.
5. Plant Cell Turgor Pressure
When water molecules move into plant cells:
- Internal pressure builds
- Cells become rigid
This pressure helps plants maintain structure and stand upright.
Osmotic Pressure: The Force Driving Water Flow
Osmotic pressure refers to the pressure required to stop the net movement of water across a semipermeable membrane.
It depends on:
- Solute concentration
- Number of solute particles
- Temperature
When higher solute concentration exists on one side of the membrane, water flows toward that side until equilibrium is reached.
Thus, osmotic pressure controls the movement of water across biological membranes.
List of Experiments on Osmosis
Osmosis experiments help students observe the movement of water across a semipermeable membrane and understand how water moves from a higher concentration of water to a lower water concentration. These experiments are widely used in biology laboratories to demonstrate what happens inside the cell when the surrounding environment changes.
Below is a list of common laboratory and classroom experiments used to study osmosis.
1. Potato Osmosis Experiment
Objective:
To observe how water moves in plant tissues through osmosis.
Procedure overview:
- Cut potato strips of equal size.
- Place them in different salt or sugar solutions.
- Leave one strip in distilled water (high water concentration).
- Measure length or mass before and after the experiment.
Observation:
- Potato strips placed in distilled water swell because water moves inside the cell from a higher concentration of water outside.
- Strips placed in salt solution shrink due to lower water concentration outside the cells.
Concept demonstrated:
Water moves from higher concentration to lower water concentration across the cell membrane.
2. Dialysis Tubing Osmosis Experiment
Objective:
To demonstrate osmosis using an artificial semipermeable membrane.
Procedure overview:
- Fill dialysis tubing with sugar solution.
- Tie the ends and place it in distilled water.
- Measure the mass or volume after a period of time.
Observation:
- Water moves into the tubing because the surrounding water has a higher concentration than the solution inside.
- The tubing swells as water moves inside the cell-like membrane.
Concept demonstrated:
Water crosses a semipermeable membrane from higher concentration to lower water concentration.
3. Red Blood Cell Osmosis Experiment
Objective:
To observe osmotic effects in animal cells.
Procedure overview:
- Place red blood cells in three solutions:
- distilled water
- isotonic saline solution
- concentrated saline solution
Observation:
- In distilled water, cells swell as water enters inside the cell.
- In concentrated saline, cells shrink due to lower water concentration outside.
- In isotonic solution, cells remain normal.
Concept demonstrated:
The direction of water movement depends on differences in higher concentration or lower water concentration between the inside and outside of cells.
4. Egg Osmosis Experiment (Decalcified Egg)
Objective:
To demonstrate osmosis using a real biological membrane.
Procedure overview:
- Soak a raw egg in vinegar to remove the shell.
- Place the egg in corn syrup or sugar solution.
- Later transfer the egg into water.
Observation:
- In syrup, water leaves the egg due to lower water concentration outside.
- In water, the egg swells because water moves inside the cell membrane from a higher concentration.
Concept demonstrated:
Osmotic water movement through a biological membrane.
5. Osmosis Using Plant Cells (Onion Cells)
Objective:
To observe plasmolysis in plant cells.
Procedure overview:
- Prepare onion epidermal cells on a microscope slide.
- Add salt solution.
- Observe under a microscope.
Observation:
- The cytoplasm shrinks away from the cell wall as water leaves the cell due to lower water concentration outside.
Concept demonstrated:
Osmotic water loss from plant cells.
6. Osmosis with Gelatin Cubes
Objective:
To study diffusion and osmosis in semi-solid structures.
Procedure overview:
- Prepare gelatin cubes containing sugar solution.
- Place them in distilled water.
Observation:
- Water enters the cubes from the surrounding solution with higher concentration of water.
Concept demonstrated:
Water movement across semi-permeable structures.
7. Reverse Osmosis Demonstration
Objective:
To demonstrate water purification principles.
Procedure overview:
- Apply pressure to force water through a semipermeable membrane.
Observation:
- Water moves against the normal osmotic gradient from lower water concentration toward a higher concentration region when pressure is applied.
Concept demonstrated:
Reverse osmosis used in desalination and water filtration.
These experiments allow students to visualize the osmosis process and understand how water moves from higher concentration areas to regions with lower water concentration across membranes. Observing what happens inside the cell during these experiments helps explain important biological processes in plants, animals, and microorganisms.
If you need help writing detailed lab reports, research papers, or experiment analysis on osmosis, the experts at IvyResearchWriters.com can assist you with professionally written academic content tailored to your course requirements.
Significance of Osmosis in Biology and Physiology
The significance of osmosis is enormous in biological systems.
Key roles include:
- Maintaining cell shape
Osmosis keeps cells balanced in isotonic solution. - Nutrient absorption in plants
Plants obtain water from the soil through osmosis. - Regulating body fluids
Human organs rely on diffusion and osmosis to regulate fluid levels. - Cell survival
Without osmosis, water molecules move uncontrollably, disrupting cell functions.
Because of these functions, osmosis is essential and important in biology.
Applications of Osmosis: From Biology to Technology
The applications of osmosis extend beyond biology into engineering and medicine.
Reverse Osmosis
Reverse osmosis forces water across a semipermeable membrane against natural osmotic flow.
Uses include:
- Water purification
- Desalination of seawater
- Laboratory filtration systems
Forward Osmosis
Forward osmosis uses natural osmotic pressure to move water across membranes.
Applications include:
- Wastewater treatment
- Food processing
- Biomedical filtration systems
Both technologies rely on the movement of water across a membrane and demonstrate practical osmosis examples.
Why Osmosis Matters in Life and Science
The osmosis process is one of the most fundamental biological mechanisms. It describes how water molecules move across a semipermeable membrane from low solute concentration to high solute concentration.
Through osmosis examples, we see how:
- Cells maintain equilibrium
- Plants absorb water
- Red blood cells respond to solution changes
- Biological systems regulate fluids
Because osmosis is essential, understanding diffusion and osmosis helps explain many processes in physiology, medicine, and environmental science.
Get Expert Help with Biology Assignments
If you need assistance writing a biology paper on osmosis examples, diffusion and osmosis, or other complex biological topics, the experts at IvyResearchWriters.com are ready to help.
Our professional academic writers can help you:
- Develop high-quality research papers
- Understand complex biological concepts
- Produce plagiarism-free academic work
Order your custom research paper today and improve your academic performance with IvyResearchWriters.com.
Frequently Asked Questions
What is osmosis in our daily life?
Osmosis appears in many everyday biological and environmental processes. In simple terms, osmosis is the movement of water across a semipermeable membrane from an area with more water to an area where water is less available because of dissolved substances.
In daily life, this process can be observed in several situations:
- Plant roots absorbing water: Water from the soil enters root cells because the soil often has a higher concentration of water than inside the cell, where dissolved nutrients reduce water availability.
- Food preservation with salt: When salt is added to foods like fish or vegetables, the environment outside microorganisms has a lower water concentration, causing water to leave microbial cells and preventing spoilage.
- Human body fluid balance: In body tissues, water moves between cells and blood depending on differences in lower concentration of water or solutes across cell membranes.
For students studying biology or writing research papers on topics such as osmosis, physiology, or cell biology, IvyResearchWriters.com provides expert academic support to help explain these concepts clearly and accurately.
What is the simplest osmosis example?
The simplest osmosis example involves a cell placed in water.
Consider the following explanation:
- When a cell membrane separates pure water from a cell containing dissolved substances, water moves from the side with higher concentration of water toward the side with lower water concentration.
- This movement happens inside the cell through the semipermeable membrane.
A common classroom example is a red blood cell placed in distilled water:
- The surrounding solution has a higher concentration of water.
- The interior of the cell has dissolved solutes, creating a lower concentration of water inside the cell.
- Water therefore enters the cell through osmosis.
Understanding such basic examples helps students grasp complex biological mechanisms. If you need help writing a biology assignment or research paper about these concepts, IvyResearchWriters.com offers professional academic writing assistance.
What are the three types of osmosis?
The three types of osmosis are determined by the relative solute and water concentrations surrounding a cell.
1. Hypotonic condition
- The surrounding solution has a higher concentration of water.
- The cell interior has a lower concentration of water because of dissolved solutes.
- Water moves inside the cell, causing the cell to swell.
2. Hypertonic condition
- The surrounding solution has lower water concentration than the cell interior.
- Water moves out of the cell from the region with higher concentration of water to the area with lower water concentration.
- The cell may shrink as water leaves.
3. Isotonic condition
- Water concentrations are balanced both outside and inside the cell.
- Although water molecules move in both directions, there is no overall change because the higher concentration and lower concentration of water differences are minimal.
Understanding these three conditions is fundamental in physiology and cell biology, and detailed explanations can be developed in research assignments with the help of the professional writers at IvyResearchWriters.com.
What is osmosis in simple terms?
In the simplest terms, osmosis is the movement of water through a semipermeable membrane from an area with more water to an area with less water.
This means:
- Water moves from a place with higher concentration of water
- Toward a place with lower water concentration
- Often moving inside the cell when the cell interior contains more dissolved substances
Because solutes reduce available water, the region with more dissolved particles has a lower concentration of water, which attracts water molecules across the membrane.
Osmosis is a fundamental concept in biology, physiology, and medicine. If you are studying these topics and need well-researched academic papers, lab reports, or explanations, IvyResearchWriters.com provides reliable academic writing services tailored to student needs.

